25 Degrees C In Kelvin
monicres
Sep 07, 2025 · 5 min read
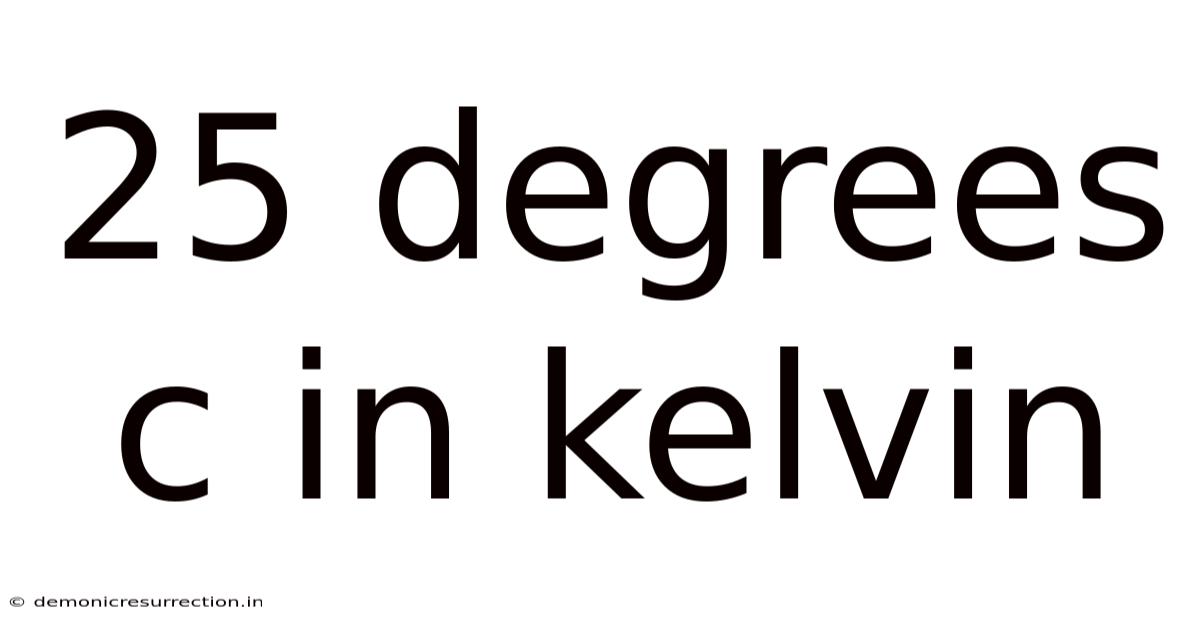
Table of Contents
25 Degrees Celsius in Kelvin: A Comprehensive Guide to Temperature Conversion
Understanding temperature conversion is crucial in many scientific fields, from physics and chemistry to meteorology and engineering. This comprehensive guide will delve into the conversion of 25 degrees Celsius (°C) to Kelvin (K), explaining the process, the underlying scientific principles, and the practical applications of this conversion. We'll also explore frequently asked questions and provide a deeper understanding of the Kelvin scale itself.
Introduction: Celsius, Kelvin, and the Importance of Conversion
The Celsius (°C) scale, also known as the centigrade scale, is a widely used temperature scale based on the freezing and boiling points of water at 0°C and 100°C, respectively, at standard atmospheric pressure. However, for many scientific applications, the Kelvin (K) scale is preferred. The Kelvin scale is an absolute temperature scale, meaning it starts at absolute zero – the theoretical point where all molecular motion ceases. Understanding how to convert between Celsius and Kelvin is therefore essential for accurate scientific calculations and interpretations. This article will specifically focus on converting 25°C to Kelvin, providing a step-by-step process and a deeper understanding of the underlying principles.
The Conversion Process: From Celsius to Kelvin
The conversion from Celsius to Kelvin is remarkably straightforward. There's only one step involved:
Kelvin (K) = Celsius (°C) + 273.15
To convert 25 degrees Celsius to Kelvin, we simply add 273.15 to the Celsius value:
25°C + 273.15 = 298.15 K
Therefore, 25 degrees Celsius is equal to 298.15 Kelvin.
A Deeper Dive into the Kelvin Scale: Absolute Zero and its Significance
The Kelvin scale's significance lies in its absolute nature. Absolute zero (0 K) represents the theoretical point at which all matter has zero thermal energy. This means that atoms and molecules have minimal vibrational and translational motion. It's important to note that absolute zero is unattainable in practice, although scientists have gotten remarkably close.
The Kelvin scale's absolute nature makes it particularly useful in scientific calculations, particularly those involving:
-
Gas Laws: Many gas laws, such as the Ideal Gas Law (PV = nRT), require temperature to be expressed in Kelvin. This is because the relationships described by these laws depend on the absolute kinetic energy of gas molecules, which is directly proportional to temperature in Kelvin.
-
Thermodynamics: In thermodynamics, Kelvin is the standard unit for temperature. Concepts like entropy and enthalpy, which are fundamental to understanding energy transfer and transformations, rely on the Kelvin scale for accurate measurements and calculations.
-
Spectroscopy: Spectroscopic techniques, used to analyze the interaction of matter with electromagnetic radiation, often utilize the Kelvin scale to relate temperature to the energy levels of atoms and molecules.
-
Astrophysics and Cosmology: The study of stars, galaxies, and the universe often involves extremely high and low temperatures. The Kelvin scale provides a consistent and unambiguous measure for these extreme temperature ranges.
Practical Applications of the 25°C to 298.15 K Conversion
The conversion of 25°C to 298.15 K has numerous practical applications across various fields. For instance:
-
Chemistry: In chemical reactions, the temperature significantly influences the reaction rate. Expressing the temperature in Kelvin allows for more precise calculations of reaction kinetics and equilibrium constants. A chemist working with a reaction at 25°C would use 298.15 K in their calculations for accurate results.
-
Physics: In physics, particularly in thermodynamics and statistical mechanics, Kelvin is essential for calculations involving energy, entropy, and other thermodynamic properties. The conversion ensures consistency and accuracy in theoretical models and experimental results.
-
Meteorology: While Celsius is commonly used in weather reports, the conversion to Kelvin is crucial for meteorological modeling and forecasting, especially when dealing with atmospheric processes and energy transfer.
-
Engineering: In various engineering disciplines, particularly chemical and mechanical engineering, the Kelvin scale is used in designing systems that involve heat transfer, such as engines, power plants, and refrigeration systems. Accurate temperature readings in Kelvin are essential for optimizing efficiency and safety.
Illustrative Example: Ideal Gas Law
Let's illustrate the importance of using Kelvin in a practical example using the Ideal Gas Law: PV = nRT.
Where:
- P = Pressure
- V = Volume
- n = Number of moles of gas
- R = Ideal gas constant
- T = Temperature (in Kelvin)
Imagine you have a gas at 25°C and want to calculate its pressure. You must use the Kelvin equivalent (298.15 K) in the equation. Using Celsius directly would lead to an incorrect calculation. This exemplifies the crucial role of Kelvin in accurate scientific calculations.
Frequently Asked Questions (FAQ)
Q: Why is the Kelvin scale preferred in science over Celsius or Fahrenheit?
A: The Kelvin scale is preferred because it's an absolute scale, starting at absolute zero. This absolute reference point simplifies many scientific calculations and provides a more fundamental understanding of temperature's relationship to energy. Celsius and Fahrenheit are relative scales, meaning their zero points are arbitrary.
Q: Can I convert from Kelvin back to Celsius?
A: Absolutely! The reverse conversion is equally simple:
Celsius (°C) = Kelvin (K) - 273.15
Q: What is the significance of absolute zero?
A: Absolute zero represents the theoretical point where all molecular motion ceases. It's a fundamental concept in thermodynamics and statistical mechanics. While unattainable in practice, it serves as a crucial reference point for the Kelvin scale.
Q: Are there other temperature scales besides Celsius, Kelvin, and Fahrenheit?
A: Yes, there are other temperature scales, although they are less commonly used. These include the Rankine scale and the Réaumur scale. Each scale has its own unique definition and application.
Conclusion: The Importance of Accurate Temperature Conversion
The conversion of 25 degrees Celsius to 298.15 Kelvin is a fundamental operation in many scientific and engineering disciplines. Understanding this conversion, and the underlying principles of the Kelvin scale, is essential for accurate calculations and a deeper understanding of temperature's role in various physical and chemical processes. The absolute nature of the Kelvin scale makes it an indispensable tool for researchers and professionals working with temperature-dependent phenomena. From the Ideal Gas Law to advanced thermodynamic calculations, the use of Kelvin ensures accuracy and facilitates a more profound understanding of the natural world. Mastering this simple conversion is a key step in grasping more complex scientific concepts and applications.
Latest Posts
Related Post
Thank you for visiting our website which covers about 25 Degrees C In Kelvin . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.