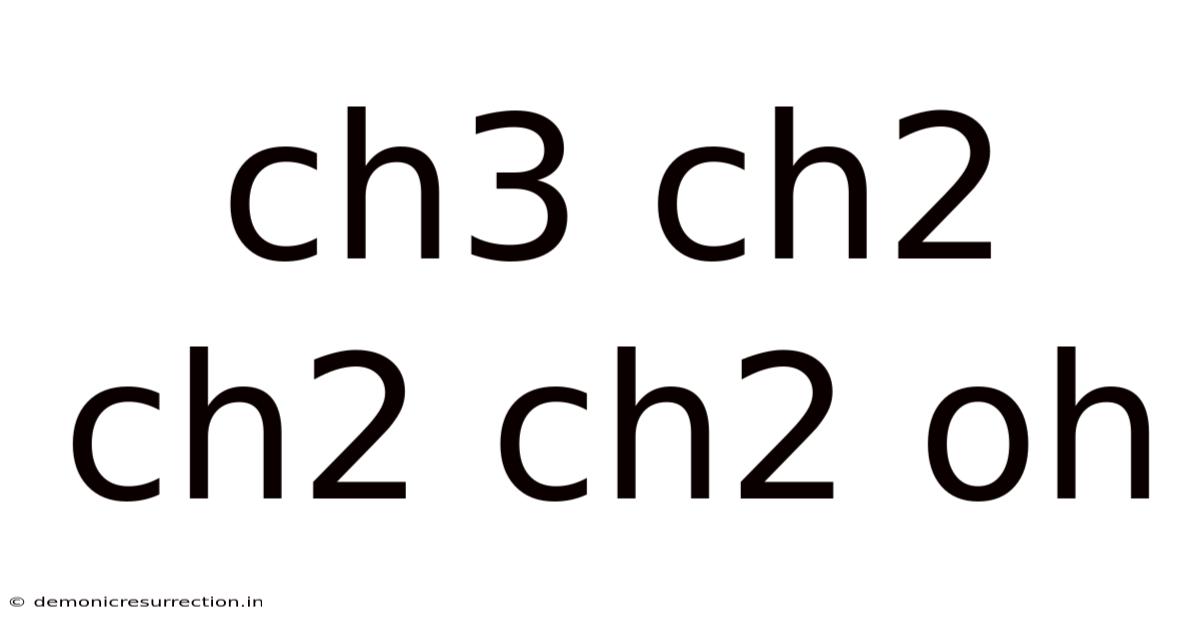Decoding the Mystery of CH3CH2CH2CH2OH: A Deep Dive into Butanol
Introduction:
What is CH3CH2CH2CH2OH? Here's the thing — for those unfamiliar with chemical notation, this seemingly cryptic string represents a molecule – specifically, n-butanol, a four-carbon alcohol with significant industrial and scientific importance. In practice, understanding CH3CH2CH2CH2OH is key to appreciating its contribution to numerous facets of modern life, from fuels to pharmaceuticals. This practical guide will unravel the mysteries behind this seemingly simple formula, exploring its properties, applications, production methods, safety considerations, and its role in various fields. We'll look at its chemical structure, explore its reactivity, and examine its diverse applications.
Understanding the Chemical Structure of n-Butanol (CH3CH2CH2CH2OH)
The chemical formula CH3CH2CH2CH2OH represents n-butanol, also known as 1-butanol. The "n" prefix stands for normal, indicating that the four carbon atoms are arranged in a straight chain. Let's break down the formula:
- CH3: This represents a methyl group, a carbon atom bonded to three hydrogen atoms.
- CH2: This represents a methylene group, a carbon atom bonded to two hydrogen atoms. There are three of these in n-butanol.
- CH2OH: This is a primary alcohol group, consisting of a carbon atom bonded to a hydroxyl group (-OH). The presence of this hydroxyl group is what classifies n-butanol as an alcohol. The location of the hydroxyl group at the end of the carbon chain is what differentiates it from other isomers of butanol.
The structure can be visualized as a chain of four carbon atoms, with the hydroxyl group attached to the terminal carbon. This linear structure contributes to n-butanol's distinct physical and chemical properties Small thing, real impact..
Physical and Chemical Properties of n-Butanol
n-Butanol possesses several key properties that determine its uses:
- Appearance: It's a colorless liquid at room temperature.
- Odor: It has a characteristic pungent, slightly alcoholic odor.
- Solubility: It's slightly soluble in water, meaning it can dissolve to a limited extent. Still, it is completely miscible with many organic solvents.
- Boiling Point: Its relatively high boiling point (117.7 °C) compared to other alcohols of similar molecular weight reflects the strength of intermolecular forces (hydrogen bonding) between molecules.
- Flammability: n-Butanol is flammable, posing a safety hazard if not handled carefully.
- Reactivity: The hydroxyl group makes n-butanol reactive. It can participate in esterification (reaction with carboxylic acids to form esters), dehydration (loss of water to form alkenes), and oxidation (conversion to butanal or butanoic acid).
Production Methods of n-Butanol
Several methods are used for the industrial production of n-butanol:
- Reppe Process: This process involves the hydroformylation of propylene, followed by hydrogenation. This method was historically important but has seen decreased usage due to the cost and safety concerns associated with high-pressure reactions.
- Fermentation: This is a bio-based production method using microorganisms like Clostridium acetobutylicum. This bacterium ferments sugars to produce a mixture of acetone, butanol, and ethanol, known as ABE fermentation. This method is gaining traction due to its renewable nature and potential for reduced environmental impact. The increasing demand for sustainable alternatives is driving interest in bio-based production of n-butanol.
- Oxoprocess: This process uses propylene as a starting material but involves a different catalytic route compared to the Reppe process, yielding n-butanol with greater efficiency.
The choice of production method depends on several factors, including cost, availability of raw materials, and environmental considerations Most people skip this — try not to..
Applications of n-Butanol
The versatility of n-butanol stems from its unique combination of properties, leading to its diverse applications across various industries:
- Solvent: n-Butanol is a widely used solvent in coatings, inks, and resins. Its ability to dissolve both polar and nonpolar substances makes it highly effective in many applications. It's often employed in the formulation of paints, varnishes, and lacquers.
- Intermediate Chemical: It serves as a key intermediate in the synthesis of many chemicals, including esters, ethers, and other valuable compounds. Its reactivity enables its use as a building block in the production of more complex molecules.
- Fuel Additive: n-Butanol is considered a promising biofuel, offering a renewable and cleaner alternative to fossil fuels. Its high energy density and compatibility with existing engine technology make it attractive for use as a gasoline additive or even as a pure fuel. Research is ongoing to improve its production efficiency and lower its cost to enhance its competitiveness in the fuel market.
- Pharmaceutical Industry: It finds application in the pharmaceutical industry as a solvent and in the production of certain drugs. Its relative safety and compatibility with biological systems are important factors in its use in this sector.
- Cosmetics and Personal Care Products: Due to its relatively low toxicity and pleasant odor, it appears in some cosmetics and personal care products. That said, it is important to remember that it should be carefully screened to avoid irritation or other negative effects depending on concentration and formulation.
Safety Considerations and Handling of n-Butanol
While n-butanol is generally considered relatively safe compared to some other industrial solvents, certain precautions should be taken:
- Flammability: It's crucial to remember its flammability and avoid open flames or ignition sources when handling it. Adequate ventilation is also essential to prevent the build-up of flammable vapors.
- Toxicity: Although considered relatively low toxicity, exposure to high concentrations can cause irritation to the eyes, skin, and respiratory tract. Appropriate personal protective equipment (PPE), including gloves, eye protection, and respiratory protection, should be used when handling it, particularly in industrial settings.
- Environmental Impact: While generally less harmful than some solvents, proper disposal methods should be followed to prevent environmental pollution. Its impact on aquatic life should also be considered, as it can cause harm at high concentrations.
- Storage: n-Butanol should be stored in tightly closed containers in a cool, dry, and well-ventilated area, away from ignition sources.
n-Butanol Isomers: Exploring the Differences
n-Butanol is only one of four isomers of butanol, meaning there are other molecules with the same chemical formula (C4H10O) but different structural arrangements. These include:
- Isobutanol (2-methyl-1-propanol): The hydroxyl group is attached to a carbon atom that is also bonded to another methyl group (CH3), giving it a branched structure.
- sec-Butanol (2-butanol): The hydroxyl group is attached to a secondary carbon atom (a carbon atom bonded to two other carbon atoms).
- tert-Butanol (2-methyl-2-propanol): The hydroxyl group is attached to a tertiary carbon atom (a carbon atom bonded to three other carbon atoms).
Each of these isomers exhibits different physical and chemical properties, influencing their respective applications. To give you an idea, tert-butanol has a lower boiling point than n-butanol due to its more compact structure, which reduces intermolecular forces Nothing fancy..
Frequently Asked Questions (FAQ)
Q: What are the main differences between n-butanol and isobutanol?
A: The primary difference lies in their molecular structure. But n-Butanol has a linear carbon chain, while isobutanol has a branched structure. This structural difference leads to variations in their physical properties, such as boiling points and solubilities, and consequently, their applications Not complicated — just consistent..
Worth pausing on this one.
Q: Is n-butanol toxic?
A: n-Butanol has relatively low toxicity compared to some other solvents. Still, exposure to high concentrations can cause irritation to the eyes, skin, and respiratory tract. Appropriate safety precautions and PPE should always be used Which is the point..
Q: What are the environmental implications of n-butanol production and use?
A: The environmental impact of n-butanol depends on the production method. Now, bio-based production via fermentation is generally considered more environmentally friendly than methods relying on fossil fuels. Proper disposal and handling practices are essential to minimize any negative environmental effects No workaround needed..
Q: What is the future outlook for n-butanol?
A: The future of n-butanol looks promising, particularly as a sustainable alternative to fossil fuels. Here's the thing — continued research and development in bio-based production methods are likely to increase its availability and reduce its cost, expanding its applications in various industries. The increasing global demand for sustainable materials will propel its growth.
Conclusion
n-Butanol (CH3CH2CH2CH2OH), with its versatile properties and diverse applications, is a crucial chemical compound with a significant role in modern society. From its use as a solvent and intermediate in chemical synthesis to its potential as a biofuel, n-butanol's importance is undeniable. Understanding its chemical structure, properties, production methods, and safety considerations is vital for harnessing its potential while mitigating any potential risks. As research continues to improve its sustainable production and broaden its applications, n-butanol is set to play an even more prominent role in the future. Its unique blend of properties makes it a key player in numerous industries and will continue to be a valuable resource for years to come. The transition towards more sustainable and environmentally friendly solutions will only amplify the demand for this important chemical compound.