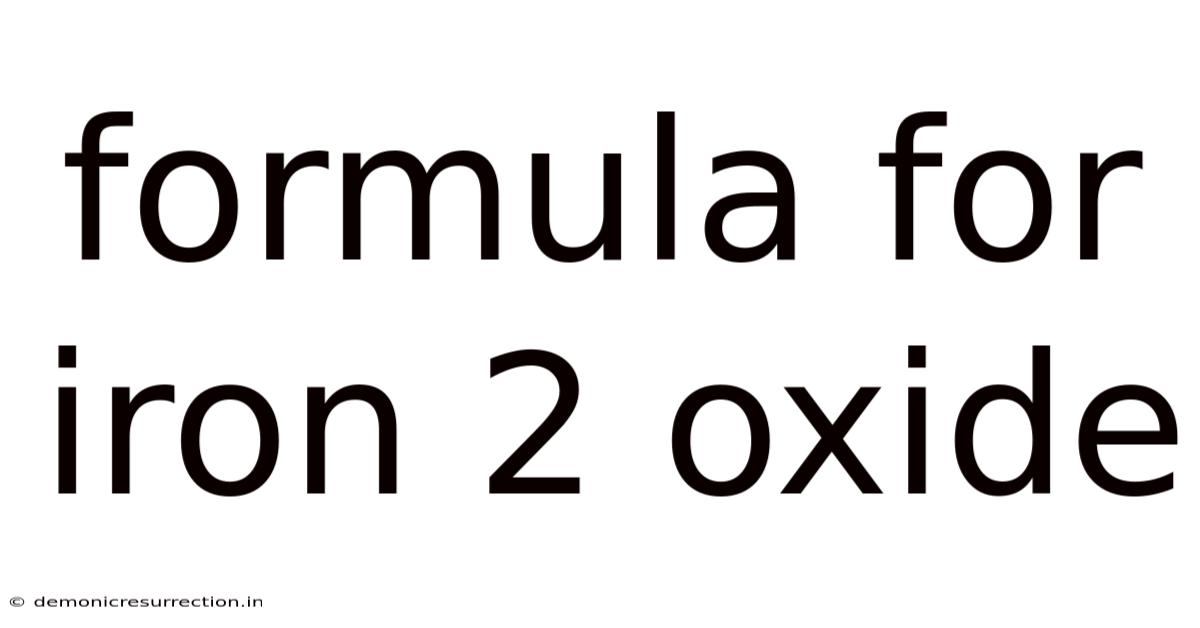Unveiling the Formula and Fascinating World of Iron(II) Oxide
Iron(II) oxide, also known as ferrous oxide, is a chemical compound with a rich history and a wide range of applications. That's why understanding its formula, properties, and uses is crucial for anyone interested in chemistry, materials science, or even history. This comprehensive article delves deep into the world of iron(II) oxide, explaining its formula, exploring its diverse applications, and addressing frequently asked questions That's the part that actually makes a difference. No workaround needed..
Some disagree here. Fair enough.
Introduction: Decoding the Formula FeO
The formula for iron(II) oxide is simply FeO. Here's the thing — this seemingly simple formula encapsulates a wealth of chemical information. And the "Fe" represents the element iron (Ferrum), and the "O" represents oxygen. The Roman numeral (II) or the prefix "ferrous" indicates that iron is in its +2 oxidation state. On top of that, this means each iron atom has lost two electrons, resulting in a balanced charge with the oxygen atom which has a -2 oxidation state. This balance is crucial for the compound's stability. Understanding this simple formula is the key to unlocking the complex properties and behaviors of this important compound.
Understanding the Chemical Bonding in FeO
The bonding in iron(II) oxide is primarily ionic. What this tells us is iron atoms donate their valence electrons to oxygen atoms, forming positively charged iron(II) ions (Fe²⁺) and negatively charged oxide ions (O²⁻). On top of that, the electrostatic attraction between these oppositely charged ions holds the crystal lattice together. While predominantly ionic, there's a degree of covalent character to the bond, meaning some electron sharing also occurs. The degree of ionic versus covalent character depends on factors like electronegativity differences and crystal structure.
Most guides skip this. Don't.
Preparation and Synthesis of Iron(II) Oxide
Several methods exist for synthesizing iron(II) oxide, each with its own advantages and disadvantages:
-
Controlled Oxidation of Iron: One common method involves carefully heating iron metal in a controlled oxygen atmosphere. The reaction must be controlled precisely to prevent the formation of higher oxidation states of iron, such as Fe₃O₄ (magnetite) or Fe₂O₃ (hematite). This method yields a relatively pure sample of FeO. The reaction is represented as:
2Fe(s) + O₂(g) → 2FeO(s) -
Reduction of Iron(III) Oxide: Iron(III) oxide (Fe₂O₃) can be reduced using various reducing agents, such as hydrogen gas or carbon monoxide, at high temperatures. This process involves a careful balance of temperature and reducing agent concentration to achieve the desired iron(II) oxide product. An example reaction using hydrogen is:
Fe₂O₃(s) + H₂(g) → 2FeO(s) + H₂O(g) -
Decomposition of Iron(II) Oxalate: Heating iron(II) oxalate (FeC₂O₄) in the absence of air also produces iron(II) oxide. This method offers a relatively pure product but requires careful control of the heating process to avoid the formation of unwanted byproducts. The reaction is:
FeC₂O₄(s) → FeO(s) + CO(g) + CO₂(g)
Each synthesis method yields iron(II) oxide with slightly varying properties depending on factors such as particle size, purity, and crystal structure.
Properties of Iron(II) Oxide: A Detailed Examination
Iron(II) oxide exhibits several key properties that contribute to its diverse applications:
-
Appearance: Pure FeO is typically a black, crystalline solid. Still, it's often found in various shades of gray, brown, or even green depending on impurities and the method of preparation That's the whole idea..
-
Crystal Structure: FeO crystallizes in a rock salt structure (NaCl structure), a type of cubic crystal system. This arrangement features a regular arrangement of Fe²⁺ and O²⁻ ions in a three-dimensional lattice Which is the point..
-
Magnetic Properties: While not ferromagnetic like iron metal, FeO exhibits antiferromagnetic properties below a certain temperature (the Néel temperature). This means the magnetic moments of neighboring iron atoms align in opposite directions, resulting in a net magnetic moment of zero Small thing, real impact..
-
Reactivity: Iron(II) oxide is relatively reactive, especially in the presence of oxygen or acids. It readily oxidizes to form higher oxidation states of iron, particularly Fe₃O₄. Reactions with acids result in the formation of iron(II) salts.
-
Solubility: Iron(II) oxide is only slightly soluble in water but readily dissolves in acids.
Applications of Iron(II) Oxide: A Multifaceted Compound
The diverse properties of iron(II) oxide contribute to its widespread applications in various industries:
-
Pigments and Colorants: Iron(II) oxide, particularly in its various hydrated forms, is used as a pigment in paints, ceramics, and other materials. It provides a range of colors, from black to various shades of brown and green, depending on the method of preparation and the presence of impurities Easy to understand, harder to ignore. And it works..
-
Ceramics and Glass Manufacturing: It's used in the manufacturing of ceramics and glass to control color and properties. It can act as a coloring agent or a flux, influencing the melting point and viscosity of the molten materials.
-
Catalysis: Iron(II) oxide acts as a catalyst in certain chemical reactions, notably in the synthesis of ammonia and in various oxidation-reduction reactions.
-
Steel Production: While not a direct component of steel, iron(II) oxide plays a role in steelmaking processes through its influence on the overall oxidation-reduction reactions.
-
Magnetic Materials: Though not directly used in strong magnets, its antiferromagnetic properties are studied and have implications for the development of new magnetic materials.
-
Mineral Supplements: Iron(II) oxide is a source of iron and is used in some mineral supplements.
FAQ: Addressing Common Questions about Iron(II) Oxide
-
Is iron(II) oxide toxic? While not highly toxic, ingestion of large quantities can lead to health issues. Appropriate handling and safety precautions should always be followed Less friction, more output..
-
What is the difference between iron(II) oxide and iron(III) oxide? The key difference lies in the oxidation state of iron. Iron(II) oxide (FeO) contains iron in the +2 oxidation state, while iron(III) oxide (Fe₂O₃) contains iron in the +3 oxidation state. This difference leads to distinct properties and applications.
-
How is iron(II) oxide identified? Identification can involve several techniques such as X-ray diffraction (XRD) to determine its crystal structure, and chemical analysis to confirm its composition and oxidation state. Visual inspection alone is not sufficient for accurate identification due to its varied appearance depending on purity and preparation Easy to understand, harder to ignore..
-
Is iron(II) oxide stable? Pure iron(II) oxide is not particularly stable in air, readily oxidizing to form Fe₃O₄ (magnetite). it helps to store it under inert conditions to maintain its purity.
-
Can iron(II) oxide be used in everyday life? While not directly seen in many everyday applications, it's a component in several products we use daily, such as pigments in paints and coatings.
Conclusion: A Vital Compound with Expanding Applications
Iron(II) oxide, with its simple formula FeO, holds a significant position in chemistry and various industrial applications. Its unique properties, stemming from the +2 oxidation state of iron and its rock salt crystal structure, contribute to its diverse uses in pigments, ceramics, catalysis, and even mineral supplements. Which means this detailed exploration hopefully provides a comprehensive understanding of this fascinating chemical compound and its significant role in the world around us. Further research into its properties and potential applications continues to expand its importance in scientific and industrial fields Easy to understand, harder to ignore. That's the whole idea..